|
| 1 |
For a list of common ions see:
http://murov.info/ions.htm
What is the name of SO42- ? |
|
|
|
sulfide |
|
|
|
sulfite |
|
|
|
sulfate |
|
|
|
sulfur tetroxide |
|
|
|
None of the previous answers. |
|
| 2 |
What is the name of NO2-
? |
|
|
|
nitrite |
|
|
|
nitrogen dioxide |
|
|
|
nitrate |
|
|
|
nitrogen peroxide |
|
|
|
None of the previous answers. |
|
| 3 |
What is the name of OH- ? |
|
|
|
Hydrox |
|
|
|
hydride |
|
|
|
oxide |
|
|
|
hydroxide |
|
|
|
None of the previous answers. |
|
| 4 |
What is the name of CO32-
? |
|
|
|
carbonite |
|
|
|
bicarbonate |
|
|
|
oxalate |
|
|
|
chromate |
|
|
|
None of the previous answers. |
|
| 5 |
What is the name of NH4+
? |
|
|
|
ammonia |
|
|
|
ammonium |
|
|
|
nitrogen tetrahydride |
|
|
|
nitrate |
|
|
|
None of the previous answers. |
|
| 6 |
What is the name of SO32-
? |
|
|
|
sulfite |
|
|
|
sulfate |
|
|
|
sulfide |
|
|
|
sulfur trioxide |
|
|
|
none of the previous answers. |
|
| 7 |
What is the name of ClO3-
? |
|
|
|
chlorite |
|
|
|
chlorate |
|
|
|
perchlorate |
|
|
|
chlorine trioxide |
|
|
|
None of the previous answers. |
|
| 8 |
What is the name of HCO3-
? |
|
|
|
bioxalate |
|
|
|
carbonic acid |
|
|
|
hydrogen carbonate |
|
|
|
hydrogen chromate |
|
|
|
None of the previous answers. |
|
| 9 |
What is the name of H3O+
? |
|
|
|
water |
|
|
|
oxygenate |
|
|
|
hydroxide |
|
|
|
hydronium ion |
|
|
|
None of the previous answers. |
|
| 10 |
What is the name of NO3-
? |
|
|
|
nitrite |
|
|
|
nitrate |
|
|
|
nitrogen trioxide |
|
|
|
nitric acid |
|
|
|
None of the previous answers. |
|
| 11 |
What is the name of ClO2-
? |
|
|
|
chloride |
|
|
|
chlorate |
|
|
|
chlorite |
|
|
|
chlorine dioxide |
|
|
|
None of the previous answers. |
|
| 12 |
What is the name of C2O42-
? |
|
|
|
carbonate |
|
|
|
chromate |
|
|
|
dicarbonite |
|
|
|
bicarbonate |
|
|
|
None of the previous answers. |
|
| 13 |
What is the name of CN- ? |
|
|
|
nitride |
|
|
|
carbide |
|
|
|
cyanide |
|
|
|
carbon nitride |
|
|
|
None of the previous answers. |
|
| 14 |
What is the name of S2O32-
? |
|
|
|
sulfate |
|
|
|
sulfite |
|
|
|
disulfate |
|
|
|
thiosulfate |
|
|
|
None of the previous answers. |
|
| 15 |
What is the name of SCN- ? |
|
|
|
cyanide |
|
|
|
cyanate |
|
|
|
thiocyanate |
|
|
|
thiosulfate |
|
|
|
None of the previous answers. |
|
| 16 |
What is the name of MnO4-
? |
|
|
|
permanganate |
|
|
|
manganate |
|
|
|
manganese tetroxide |
|
|
|
magnesium tetroxide |
|
|
|
None of the previous answers. |
|
| 17 |
What is the name of PO43-
? |
|
|
|
phosphite |
|
|
|
phosphate |
|
|
|
perphosphate |
|
|
|
phosphorous tetroxide |
|
|
|
None of the previous answers. |
|
| 18 |
What is the name of CrO42-
? |
|
|
|
chromate |
|
|
|
carbonate |
|
|
|
oxalate |
|
|
|
dichromate |
|
|
|
None of the previous answers. |
|
| 19 |
What is the name of HSO4-
? |
|
|
|
hydrogen sulfite |
|
|
|
sulfate |
|
|
|
bisulfate |
|
|
|
sulfuric acid |
|
|
|
None of the previous answers. |
|
| 20 |
What is the name of IO3-
? |
|
|
|
iodite |
|
|
|
iodide |
|
|
|
iodate |
|
|
|
periodate |
|
|
|
None of the previous answers. |
|
| 21 |
Carbonate is: |
|
|
|
C2O42- |
|
|
|
CO32- |
|
|
|
CO3- |
|
|
|
CO22- |
|
|
|
None of the previous answers. |
|
| 22 |
Oxalate is: |
|
|
|
C2O42- |
|
|
|
CO32- |
|
|
|
C2O4- |
|
|
|
C2O52- |
|
|
|
None of the previous answers. |
|
| 23 |
Chromate is: |
|
|
|
CrO32- |
|
|
|
CO32- |
|
|
|
Cr2O72- |
|
|
|
CrO42- |
|
|
|
None of the previous answers. |
|
| 24 |
Bicarbonate is: |
|
|
|
HCO3- |
|
|
|
CO32- |
|
|
|
C2O62- |
|
|
|
C2O42- |
|
|
|
None of the previous answers. |
|
| 25 |
Sulfite is: |
|
|
|
SO42- |
|
|
|
S2O32- |
|
|
|
SO32- |
|
|
|
SO3- |
|
|
|
None of the previous answers. |
|
| 26 |
Bisulfate is: |
|
|
|
HSO32- |
|
|
|
HSO4- |
|
|
|
S2O82- |
|
|
|
SO32- |
|
|
|
None of the previous answers. |
|
| 27 |
Thiosulfate is: |
|
|
|
SO42- |
|
|
|
S2O32- |
|
|
|
SO32- |
|
|
|
SCN- |
|
|
|
None of the previous answers. |
|
| 28 |
Sulfate is: |
|
|
|
HSO4- |
|
|
|
S2O32- |
|
|
|
SO32- |
|
|
|
SO4- |
|
|
|
None of the previous answers. |
|
| 29 |
Thiocyanate is: |
|
|
|
SCN- |
|
|
|
S2O32- |
|
|
|
SO32- |
|
|
|
SCN2- |
|
|
|
None of the previous answers. |
|
| 30 |
Nitrate is: |
|
|
|
NO2- |
|
|
|
NO3- |
|
|
|
NO32- |
|
|
|
NO4- |
|
|
|
None of the previous answers. |
|
| 31 |
Nitrite is: |
|
|
|
NO2- |
|
|
|
NO3- |
|
|
|
NO22- |
|
|
|
NO2 |
|
|
|
None of the previous answers. |
|
| 32 |
Phosphate is: |
|
|
|
PO33- |
|
|
|
PO42- |
|
|
|
PO43- |
|
|
|
PO32- |
|
|
|
None of the previous answers. |
|
| 33 |
Chlorate is: |
|
|
|
ClO32- |
|
|
|
ClO4- |
|
|
|
ClO2- |
|
|
|
ClO3- |
|
|
|
None of the previous answers. |
|
| 34 |
Chlorite is: |
|
|
|
ClO22- |
|
|
|
ClO2- |
|
|
|
ClO3- |
|
|
|
ClO- |
|
|
|
None of the previous answers. |
|
| 35 |
Hypochlorite is: |
|
|
|
ClO2- |
|
|
|
HClO2 |
|
|
|
ClO2- |
|
|
|
ClO- |
|
|
|
None of the previous answers. |
|
| 36 |
Ammonium is: |
|
|
|
NH3 |
|
|
|
NH4+ |
|
|
|
NH42+ |
|
|
|
NH2- |
|
|
|
None of the previous answers. |
|
| 37 |
Hydronium is: |
|
|
|
H3O2+ |
|
|
|
H2O+ |
|
|
|
H3O+ |
|
|
|
HO- |
|
|
|
None of the previous answers. |
|
| 38 |
Hyrdoxide is: |
|
|
|
A cookie. |
|
|
|
O2- |
|
|
|
H3O+ |
|
|
|
OH- |
|
|
|
None of the previous answers. |
|
| 39 |
Cyanide is: |
|
|
|
CN- |
|
|
|
NCO- |
|
|
|
N3- |
|
|
|
SCN- |
|
|
|
None of the previous answers. |
|
| 40 |
The negative ions of the elements end with the letters: |
|
|
|
ite |
|
|
|
ate |
|
|
|
ide |
|
|
|
ine |
|
|
|
None of the previous answers. |
|
| 41 |
Chlorite, nitrite and sulfite: |
|
|
|
have one more oxygen than chlorate, nitrate and sulfate. |
|
|
|
have one less oxygen than chlorate, nitrate and sulfate. |
|
|
|
are the negative ions of the elements. |
|
|
|
all have two oxygens. |
|
|
|
all have a negative one charge. |
|
| 42 |
For thiosulfate and thiocyanate, the prefix means: |
|
|
|
three |
|
|
|
the ion has a negative 3 charge |
|
|
|
an oxygen has been replaced by a sulfur |
|
|
|
None of the previous answers. |
|
|
|
|
|
| 43 |
Polyatomic ions that have the formula XOnm- where X is the central atom (such as C, N, S, P, Cl, Cr) and n and m are whole numbers that depend on the nature of X and its oxidation number are also called oxoanions or oxyanions. For commonly occurring oxoanions, the X in XO32- could be: |
|
|
|
S |
|
|
|
N |
|
|
|
P |
|
|
|
Cl |
|
|
|
None of the previous answers. |
|
| 44 |
Polyatomic ions that have the formula XOnm- where X is the central atom (such as C, N, S, P, Cl, Cr) and n and m are whole numbers that depend on the nature of X and its oxidation number are also called oxoanions or oxyanions. For commonly occurring oxoanions, the X in XO43- could be: |
|
|
|
S |
|
|
|
N |
|
|
|
P |
|
|
|
Br |
|
|
|
None of the previous answers. |
|
| 45 |
Polyatomic ions that have the formula XOnm- where X is the central atom (such as C, N, S, P, Cl, Cr) and n and m are whole numbers that depend on the nature of X and its oxidation number are also called oxoanions or oxyanions. For commonly occurring oxoanions, the X in XO2- could be: |
|
|
|
S |
|
|
|
C |
|
|
|
P |
|
|
|
Cl |
|
|
|
None of the previous answers. |
|
| 46 |
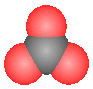 Assuming the space filling model represents an ion with a negative 2 charge, it could represent:
Assuming the space filling model represents an ion with a negative 2 charge, it could represent: |
|
|
|
carbonate |
|
|
|
sulfur trioxide |
|
|
|
sulfate |
|
|
|
nitrate |
|
|
|
None of the previous answers. |
|